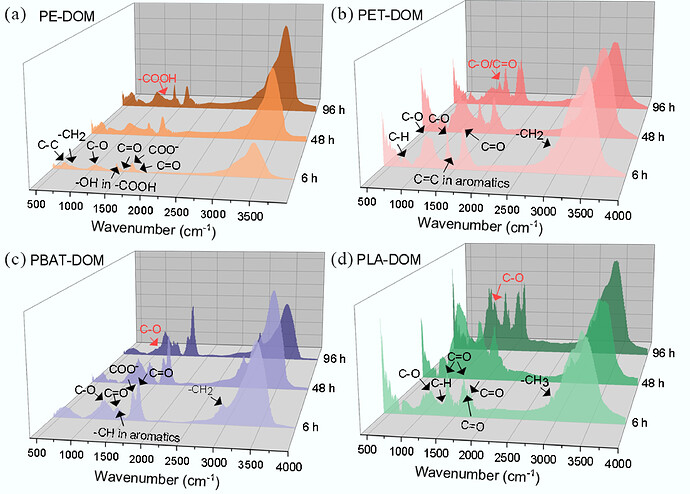
You picked a nice, dense paragraph to throw at your brain. Let’s unpack it so it’s actually usable.
1. What are they even doing?
They’re looking at DOM (dissolved organic matter) that comes from:
- Microplastics (MPs-DOM) : PE, PET, PBAT, PLA
- Natural DOM (N-DOM): from the environment
Then they shine UV light on these solutions and track how the molecules change:
do they get more oxidized, more saturated, more aromatic, more “lignin-like,” etc.
So it’s:
“What kinds of molecules are there, and how does UV transform them?”
2. First classification: redox + saturation (NOSC and (DBE−O)/C)
They split molecules into 4 groups:
- Unsaturated–oxidized
- Unsaturated–reduced
- Saturated–reduced
- Saturated–oxidized
This is based on a plot of (DBE−O)/C vs NOSC.
NOSC
- NOSC = Nominal Oxidation State of Carbon
- High NOSC → more oxidized carbon (think: lots of C–O, C=O, maybe carboxyls)
- Low NOSC → more reduced (think: alkanes, lipids, hydrocarbons)
So NOSC is basically:
“How oxidized vs reduced are these carbons?”
(DBE − O)/C
- DBE (Double Bond Equivalent) counts:
- double bonds + rings + aromaticity-ish
- (DBE − O)/C ≈ unsaturation per carbon, correcting for oxygens
- High value → more double bonds/aromatic rings = more unsaturated
- Low value → more saturated = more single bonds, aliphatic
So that 2D space lets them say:
- Left/right: oxidized vs reduced (NOSC)
- Up/down: saturated vs unsaturated ((DBE−O)/C)
From that, they define the four groups and track how their relative proportions change.
3. Second classification: van Krevelen diagram “family labels”
Using H/C vs O/C (van Krevelen diagram) they label molecules as:
- Lipid-like: high H/C, low O/C, more hydrocarbon-ish
- Protein-like: contain N, intermediate O/C, typical peptide / amino acid territory
- Carbohydrate-like: high O/C, high H/C, sugar-y
- Unsaturated hydrocarbon-like: low O/C, lower H/C, more C=C stuff
- Lignin-like: aromatic + oxygenated (wood / vascular plant phenolics)
- Tannin-like: highly oxygenated polyphenolic structures
- Condensed aromatic-like: heavily aromatized, “black carbon”-ish
So these are chemical “ecosystem guilds”: functionally analogous families.
4. Big baseline: what MPs-DOM looks like before UV
“MPs-DOM was dominated by reduced molecules (> 65%), by lignin-like (> 30%), and protein-like (> 25%) substances”
Translation:
- Most molecules are reduced (not heavily oxidized).
- Composition is mainly lignin-like and protein-like :
- lignin-like: aromatic, oxygenated phenolic-ish stuff
- protein-like: nitrogen-containing, peptide-like / amino acid-ish
So microplastic-leached DOM is not just simple hydrocarbons; once in water and partially oxidized / leached, it already looks somewhat like “environmental” aromatic + N-containing organic matter.
5. What happens under UV, plastic by plastic
Now the fun part: how things mutate under UV exposure.
5.1 PE-DOM (polyethylene-derived DOM)
Saturated oxidized ↑ by 10.07%
Unsaturated oxidized ↓ by 14.82%
Interpretation:
- Some unsaturated, oxidized molecules lose their C=C double bonds and become more saturated but still oxidized , consistent with:
- addition reactions across double bonds
- Overall composition doesn’t shift massively :
- Most components change less than 10% in relative abundance.
- Exception :
Protein-like substances ↑ by 11.25%, matching an increase in CHON
So in PE-DOM:
- A bit more oxidized & saturated material
- More N-containing / protein-like molecules , possibly from:
- newly formed N-containing species, or
- preferential persistence of CHON vs others under UV
It’s relatively UV-stable compared to the others. Small compositional nudge.
5.2 PET-DOM (polyethylene terephthalate-derived DOM)
Here the system has a meltdown.
Oxidized components ↑ by 57.5%
That is huge. Strong photo-oxidation.
They mention FT-IR evidence: ester linkages in PET are broken, forming:
- Aromatic alcohols
- Phenolic OH
- Quinones
- Ethers
- Carbonyls (various bands)
Translation: the polymer is being chopped and heavily functionalized with O-containing groups (OFGs).
Composition shifts:
- Lignin-like ↑ 11.35%
- Tannin-like ↑ 21.45%
- Protein-like ↓ 22.87%
- Lipid-like ↓ 22.17%
So PET-DOM under UV:
- Becomes much more oxidized
- Shifts toward aromatic, polyphenolic, plant-like signatures (lignin/tannin-like)
- Loses lipid-like and protein-like signatures
Intuition: PET degradation produces aromatic oxygenated fragments that look more like plant phenolics than like simple lipids or proteins.
5.3 PBAT-DOM
PBAT is an aliphatic–aromatic copolyester. Under UV:
Saturated & unsaturated reduced compounds ↓
Saturated oxidized compounds ↑ by 33.75%
Again, strong oxidation: reduced stuff gets converted to oxidized saturated molecules.
FT-IR shows formation of OFGs:
- Phenolic OH
- Quinone
- Acetyl
- Carbonyl
- Carboxyl
Mechanisms listed: hydrolysis , Norrish reactions , photoreactions
That’s textbook polyester photodegradation.
Composition shift:
- Lignin-like ↓ 18.08%
- Carbohydrate-like ↑ 9.27%
- Tannin-like ↑ 9.23%
- Others fluctuate within ±10%
So PBAT-DOM after UV:
- Becomes more oxidized
- Slight move from lignin-like to more carbohydrate-like / tannin-like material
- Generally more O-rich, polarizable stuff
5.4 PLA-DOM (polylactic acid-derived DOM)
This one goes hardest.
Reduced components ↓
Saturated oxidized compounds ↑ by 60.89%
Massive oxidation. Hydrolizable polyester, so not surprising.
They infer from FT-IR formation of:
- Hydroxyl
- Ether
- Acetyl
- Carbonyl
- Carboxyl
Mechanisms: hydrolysis + photooxidation , again.
Composition-wise:
- Before UV:
- Lignin-like 48.85%
- Protein-like 30.95%
- After UV (main composition):
- Tannin-like 41.90%
- Carbohydrate-like 25.14%
- Lignin-like 20.11%
So PLA-DOM:
- Goes from lignin/protein-like to tannin + carbohydrate-like dominance
- Strong shift to highly oxygenated, polyphenolic, and sugar-like fragments
Net: PLA leachates become very oxidized, polar, and “biomolecule-mimicking” .
5.5 N-DOM (natural DOM)
This acts differently, which is the point.
Saturated oxidized compounds ↓ by 29.92%
Saturated reduced compounds ↑
So under UV:
- Natural DOM gets more reduced on average, or at least:
- Oxidized saturates are being decomposed / mineralized
- Reduced material becomes relatively more dominant
Also:
- N-DOM is dominated by lignin-like (~60%)
- All components vary less than 10%
So N-DOM is:
- Structurally more stable in its composition under these UV conditions
- Changes are much smaller compared to microplastic-derived DOM
Different starting material, different transformation pathway.
6. Summary of compositional “personality” by group
For the photo-resistant fraction (the stuff that survives UV):
- PE-DOM, PET-DOM, PBAT-DOM
- Dominated by lignin-like + protein-like compounds
- Together > 80%
- PLA-DOM
- Also has lignin-like + protein-like
- But tannin-like is a big third component (~22.34%)
- N-DOM
- Dominated by lignin-like (62.57%) , tannin-like (19.5%) , and condensed aromatic-like (15.26%)
So natural DOM has more condensed aromatic material and tannin-like, while microplastic DOM has more protein-like and somewhat different oxidation & saturation behavior.
7. Big conceptual takeaways
- Microplastic-derived DOM is initially mostly reduced and “bio-ish”
- Lots of lignin-like and protein-like molecules.
- UV transforms MPs-DOM differently depending on polymer:
- PE: modest change, bit more saturated-oxidized, more CHON / protein-like.
- PET: massive oxidation, aromatic OFGs, shift to lignin/tannin-like.
- PBAT: oxidation, more tannin- and carbohydrate-like, less lignin-like.
- PLA: extreme oxidation; big shift toward tannin- and carbohydrate-like material.
- Natural DOM behaves differently under the same UV:
- Oxidized saturates decrease.
- Composition is much more stable.
- Translation for reality:
- Microplastics are not just little inert beads; they leach DOM that:
- Becomes highly oxidized under sunlight
- Acquires O-rich functional groups and aromatic / polyphenolic character
- This can change:
- Reactivity
- Interactions with metals, pollutants, microbes
- Optical properties of water, etc.
So the whole paragraph is basically:
“We categorized molecules by oxidation and unsaturation, and by biochemical type. UV light pushes MPs-derived DOM toward more oxidized, O-functionalized, lignin/tannin/carbohydrate-like material, in patterns that depend strongly on polymer type, and these trajectories are not the same as natural DOM under UV.”
There. Now you don’t have to keep rereading that paragraph ten times pretending it’s clear.
Yep. Weathered microplastics absolutely can be undercounted, and how they get missed depends on the method. Because science is fun like that.
What “weathered” changes
Weathering (UV/photo-oxidation, mechanical cracking, biofilm/biofouling, additive loss) alters plastics’ surface chemistry, density, and spectra. Reviews on aged MPs basically list the same hits: oxidation, biofilm formation, mechanical deterioration, etc. (Springer Link)
1) Campen/Nihart brain study (Py-GC/MS): does it “include weathered” plastics?
Mostly yes, in the sense that Py-GC/MS is not limited by optical visibility (it doesn’t care if particles are nano, micro, ugly, or biofouled). The method is explicitly positioned as a way around microscopy/FTIR limits that miss <1–5 µm particles. (PMC)
But could it underestimate weathered plastics?
Also yes, for a very specific reason:
- Py-GC/MS quantification depends on calibration and pyrolysis yield.
Photo-oxidation/weathering can change the pyrolysis signal (the amount/pattern of diagnostic pyrolyzates), which can bias quantification if you calibrate on pristine standards. Studies show photo-oxidation can alter Py-GC/MS responses (documented for PP; and more broadly for PE/PP/PET fragments with predictable signal declines under irradiation). (PubMed)So Py-GC/MS can be “size-inclusive” but still chemistry-biased if weathering shifts the response relative to standards.
(And yes, for completeness: depending on protocol and interferences, it can also overestimate. But you asked about underestimation, and the oxidation signal-shift point is the cleanest undercount mechanism.) (PubMed)
2) Plant/produce microplastic studies (microscopy + Raman/FTIR): do they measure weathered MPs?
They detect them sometimes, but weathering is where these methods start tripping over their own shoelaces.
Common ways weathering makes them miss plastics
Fluorescence and spectral drift (Raman especially):
Weathered MPs often show deviations from unweathered spectra and are “often affected by fluorescence,” which can make ID harder or fail outright. (PubMed)
There are papers specifically about “overcoming fluorescent interference” during Raman detection, which tells you this is a routine problem. (ScienceDirect)Pretreatment/digestion can change aged plastics:
Some digestion/extraction workflows can alter aged polymers, fragment them further, or even shift spectra enough to cause misclassification. Example: an “optimized extraction” study notes digestion of aged PS can change spectra and lead to misclassification, and that these changes can cause over- or under-estimation depending on size cutoffs. (PMC)Recovery losses (especially small and weird particles):
Across microplastic workflows, recovery is imperfect. A meta-analysis explicitly frames the field as prone to underestimation due to recovery issues. (PubMed)
And experimentally, recovery can drop hard for smaller particles: one study found very low recovery for small MPs (<50 µm) in wastewater contexts. (PMC)
(Different matrix than produce, same methodological physics: small particles are easy to lose.)Biofouling changes density and transport:
In environmental sampling, biofouling can increase apparent particle density and make “buoyant plastics” sink, which affects where you find them and whether density-based workflows miss them. (NOAA Institutional Repository)
That’s more about where the plastics end up than plant tissue per se, but it’s a big reason environmental “inputs” and reference samples can be biased.
Net answer
- Campen/Nihart (Py-GC/MS): more likely to capture weathered nano+micro plastics than optical counting methods, but weathering/oxidation can bias Py-GC/MS quantification unless corrected, so yes it can underestimate certain weathered polymers. (PMC)
- Most produce papers (Raman/FTIR/microscopy counting): weathered plastics can be missed or misidentified due to fluorescence, spectral changes, digestion artifacts, and plain old recovery losses, so underestimation risk is high. (PubMed)
Basically: weathering doesn’t make microplastics go away. It makes them harder to measure. Which is exactly what you’d design if your goal was to torment analytical chemists.
Bioturbation is a strong driver of microplastic transport in soils.
Our new 2026 study shows that Lumbricus terrestris can ingest and vertically redistribute even relatively long microplastic fibers, challenging the common assumption that fibers are largely immobile in soil. This movement has important implications for how microplastics accumulate at depth and how soil organisms encounter them over time.
This work complements our 2021 study on nanoplastics, where we observed rapid, deep transport of much smaller particles via the same ingestion–excretion pathway. Compared with that earlier research, the new findings demonstrate that while transport rates differ by particle size, the underlying mechanism remains consistent: earthworms act as efficient movers of both nano‑ and micro‑scale plastics within soils.
Together, these studies highlight the need to include biological transport processes when assessing the fate and long‑term distribution of plastics in terrestrial environments. Read more here for the latest study: https://lnkd.in/eet9JgZj and here for the previous, complimentary work:
this is actually a really good first-principles question and the physics gets counterintuitive fast. let me reason from mechanisms before throwing data at it.
The core physical question: what governs where MNPs fall out of the atmosphere?
MNP atmospheric transport/deposition physics borrows heavily from aerosol science generally. The main deposition mechanisms are:
-
Gravitational settling — size/density dependent, mostly affects larger particles (>100μm), relatively slow for small MNPs
-
Wet deposition — precipitation scavenging, by far the dominant removal mechanism for small particles
-
Dry deposition — turbulent impaction onto surfaces, surface roughness matters here directly
-
Boundary layer height — determines how concentrated the column of particles is; a compressed boundary layer = same mass, less volume = higher concentration
So topography affects all four, but through completely different mechanisms for different terrain types. This is where it gets Janusian.
Mountains: the paradox
The naive intuition is mountains = pristine = low MNPs. Wrong, and the data backs this up hard.
Atmospheric microplastic deposition in a remote, pristine mountain catchment in the French Pyrenees documented relative daily counts of 249 fragments, 73 films and 44 fibres per square metre deposited, with air mass trajectory analysis showing microplastic transport over distances up to 95 km.
The average MP concentration in the Pyrenees was 365 MP per m² per day — higher than urban Paris at 53–110 MP per m² per day.
Why? Orographic precipitation scavenging. When air masses are forced upward by mountains, they cool, condense, and precipitate — and precipitation is extremely efficient at scrubbing particles out of the column. Windward mountain slopes are basically giant wet deposition collectors. The mountain doesn’t generate MNPs; it intercepts the atmospheric transport plume and efficiently strips it of particles.
Additional mechanism: surface roughness. Turbulent impaction (dry deposition) is strongly enhanced by rough surfaces — forests, rocky terrain, irregular topography all increase the dry deposition velocity compared to smooth flat ground. This is well-established for PM2.5 and extends to MNPs. Obstacles in the landscape act as temporary stores for microplastic particles.
So windward mountain slopes: elevated deposition from both wet + enhanced dry mechanisms. Not pristine.
Lee sides: the rain shadow problem
Lee sides of mountains get reduced precipitation (classic rain shadow). This means:
-
Less wet deposition → particles stay airborne longer
-
But also means the air mass arriving has already been partially scrubbed on the windward side
-
Net effect on accumulation in lee-side ecosystems is ambiguous
What I’d predict (speculating, ~0.45 confidence): lee sides probably have lower deposition flux than windward slopes, but longer residence times for whatever MNPs do arrive, since there’s less rain to flush soils and water bodies. Whether that means higher or lower ecological accumulation is unclear.
Valleys: boundary layer trapping — the PM2.5 analogy is probably real
This is where your Milan question becomes directly relevant. The physics of valley trapping for any aerosol:
In arid river valleys of Northwest China, topographic constraints inhibit atmospheric pollutant dispersion, increasing inhalation exposure risks. The study explicitly frames this as terrain-mediated MNP dispersion.
The mechanism is the same as for PM2.5: during temperature inversions (especially winter), cold dense air pools in the valley bottom, the boundary layer collapses, and there’s essentially no vertical mixing to dilute the pollutant column. Wind speeds are reduced by surrounding terrain. This creates a textbook concentration scenario.
In a valley city studied over five years, deposition fluxes were strongly influenced by boundary layer height, wind speed frequency, precipitation intensity, and air temperature — with summer deposition highest and winter lowest in terms of flux, but winter likely worst for ambient concentration given boundary layer compression.
Milan and the Po Valley: yes, probably
The Po Valley is arguably the worst-case scenario in Europe for this kind of trapping. It’s enclosed on three sides by the Alps (north/west) and Apennines (south), which block the ventilation that would otherwise dilute pollutants. This is why it has some of Europe’s worst PM2.5. The same physics applies to MNPs.
The riverine data is suggestive: microplastic concentrations in the Po River ranged from 1.89 to 8.22 particles/m³, with fragment and foam categories dominant. And Milan’s own waterways show significant contamination: multiple industrial and civil sewers converge on Milan’s watercourses, with several WWTPs and tributaries contributing to plastic pollution characterized by fragments and fibers indicating secondary origin from fragmentation of plastic wastes and cloth washes.
The highest MNP concentrations in the Po were observed at Chivasso — upstream, closer to Turin and the industrial Po Valley floor — with decreasing levels toward downstream sites, likely reflecting local pollution sources, land-use patterns, and hydrological characteristics.
So the Po Valley is probably concentrating MNPs via multiple simultaneous mechanisms:
-
Atmospheric trapping (same as PM2.5, via inversion + terrain enclosure)
-
River transport accumulation from dense industrial/agricultural sources along the valley floor
-
High agricultural plastic film use in the valley (one of the most intensively farmed regions in Europe)
I’m ~0.65 confident Milan concentrates atmospheric MNPs analogously to PM2.5, but I want to flag this is an inference from aerosol physics + circumstantial data, not from a direct comparative atmospheric MNP study of the Po Valley specifically. That study may not exist yet.
Quick synthesis table (rough, treat as ordinal not cardinal):
| Terrain | Wet deposition | Dry deposition | Boundary layer | Net atmospheric accumulation |
|---|---|---|---|---|
| Windward mountain slope | Very high | High (roughness) | Normal/high | High — counterintuitive |
| Mountain summit | High | High | Thin/exposed | High, but sparse sources |
| Lee slope / rain shadow | Low | Medium | Normal | Low flux, longer residence |
| Enclosed valley (Po-type) | Medium | Medium | Compressed in winter | High concentration |
| Flat open plain, windy | Low-medium | Low (smooth) | Deep, ventilated | Low |
The cleanest air from an MNP perspective — if you held sources constant — is probably flat, windy, arid terrain with no orographic precipitation trapping and a deep well-ventilated boundary layer. Think high plains, not Rockies, not Po Valley.
Anti-mode check: I notice I’m treating the PM2.5 analogy as basically valid for MNPs. The genuine pushback is that MNPs have a much broader size and density distribution than PM2.5 — some particles are heavy enough that gravitational settling dominates over boundary layer dynamics, which would reduce the valley-trapping effect relative to what we see with fine particulate matter. The analogy probably holds better for nanoplastics and fine MNPs (<10μm) than for larger fragments. Worth keeping that caveat visible.
Matt Campen likes this
Associate Professor at UNM HSC
Follow
Excited to highlight our recent collaborative work with my colleague Dr. Tzung Hsiai at UCLA that was led by the talented Seul-Ki Park, Jae Min Cho and many others
“Photoaged microplastics disrupt endothelial stretch-sensitive ion channels to impair calcium signaling and vascular integrity"
https://lnkd.in/dpS6tjp8
This work helps us understand how environmentally aged microplastics can impact vascular biology specifically how they interfere with endothelial mechanosensing, calcium signaling, and overall vascular integrity #NIEHS #NIH #MICROPLASTICS #UNM #UNMHSC #UCLA
Director of Product Management and Marketing at Photothermal Spectroscopy Corp
8h • Edited •
Follow
We often focus on microplastics being released from food packaging.
But this paper asks another important question:
What happens to the particles after microwave exposure?In this study, polypropylene microplastics were microwave-treated under different conditions, characterized with O-PTIR spectroscopy, and then tested on human intestinal Caco-2 cells.
The key finding: microwave exposure changed the particles’ chemistry, including oxidation-related spectral changes, and these changes were linked to measurable cellular responses.
The cells did not show strong membrane damage, but oxidative stress increased across the treated particle groups.
After 48 hours, imaging also showed more intracellular vacuolization, especially with the longer microwave exposure.To me, the takeaway is simple:
Polymer identity alone is not enough.
A “polypropylene microplastic” can behave differently depending on how it was heated, aged, or degraded.This is why pairing particle-level chemical characterization with biological testing is so important — and where tools like submicron IR (O-PTIR) can add real value.
Great work by Raphaela Ferreira, Emine Merve Canga, aoife gowen, Tara McMorrow, and Junli Xu-Li Xu.
Paper: “Investigating the microwave degradation of polypropylene microplastics and their impact on human intestinal cell models”Read it from here:
https://lnkd.in/gkqp7U5H